ELTE Eötvös Loránd University
1117 Budapest Pázmány Péter sétány 1/A, Hungary
room 637 extension 1108
room 638 extension 1109
room 643 extension 1909
e-mail: turanyi@chem.elte.hu
Assessing the performance of recent detailed NH3 and NH3/H2 combustion mechanisms
András György Szanthoffer, Máté Papp, Tibor Nagy,
Tamás Turányi
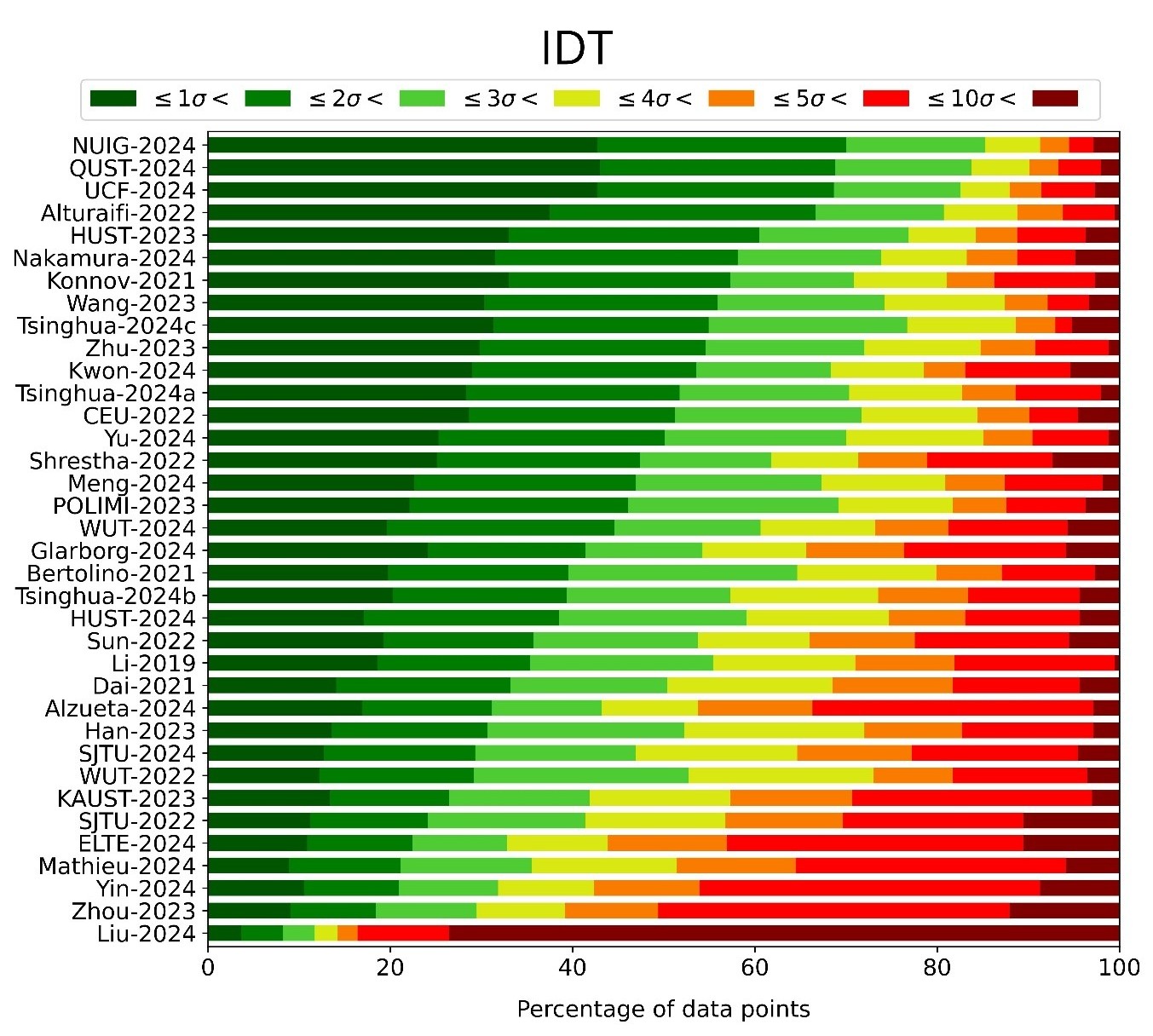
There has been growing interest in applying ammonia (NH3) as a fuel. It is often combined with hydrogen (H2), valued for being carbon-free, to compensate for the poor combustion properties of neat NH3. Over the last decade, numerous detailed NH3 combustion kinetic mechanisms have been published, but there is no consensus on the best available mechanism. This study quantitatively evaluates the performance of 36 recently published NH3 combustion mechanisms based on how well they can reproduce experimental data. The utilized experimental data collection includes shock tube ignition delay times (IDT), laminar burning velocities (LBV), and concentrations measured in flow reactors, jet stirred reactors, and shock tubes. With 17,242 data points in 1,327 data series from 110 publications, this is, to the best of our knowledge, the most extensive data collection ever used for NH3/H2 mechanism testing. The uncertainties of the experimental data were carefully evaluated, and they were considered in assessing the performance of the mechanisms. A novel, generally applicable, model-free approach was proposed to propagate the temperature uncertainty to the measured outlet concentrations, resulting in more realistic errors for the concentration data than in previous comprehensive mechanism testing studies. All utilized experimental data are available in the ReSpecTh database (https://ReSpecTh.hu). None of the mechanisms had satisfactory performance for all types of experimental data, but the NUIG 2024 mechanism (Zhu et al., 2024) and the Tsinghua 2024c mechanism (Zhang et al., 2024) demonstrated the best overall performance among the investigated models and were selected for further investigations. Missing third-body efficiencies for NH3 were added to the third-body reactions of both selected mechanisms, and their H/O and NH3 pyrolysis subsets were also replaced. The overall performance of NUIG 2024 could not be noticeably improved by these modifications, while modified versions of Tsinghua 2024c had slightly improved performance. However, none of the modifications resulted in a mechanism sufficiently accurate for all types of experimental data, which highlights the need for further NH3 combustion mechanism development.

Optima++ is a general framework for manipulating experimental data related to combustion chemistry, carrying out simulations of such experiments, performing model optimization and analysis, and providing auxiliary features for the above tasks. Optima++ is able to handle simulation codes Cantera, FlameMaster, OpenSMOKE++ and ZeroRK. Also, Chemkin Pro is coming soon.
An interactive web site, where the users may find Arrhenius parameters of gas phase elementary reactions determined in direct measurements, theoretical calculations or have been used in modelling studies. The users may recalculate the uncertainty limits of the rate coefficients. The editors have the right to upload data sheets for new reactions and to add, delete or modify existing data sheets. The editor status may be granted to any registered user upon request to the administrator.
Visit k-evaluation web page
Reaction fluxes of a combustion simulation can be visualized in the forms of still pictures and videos.
We maintain a collection of a series of Chemkin-format reaction mechanisms for the combustion of the following fuels:
hydrogen, syngas, methanol, ethanol, methane, butanol, fuels+NOx.





